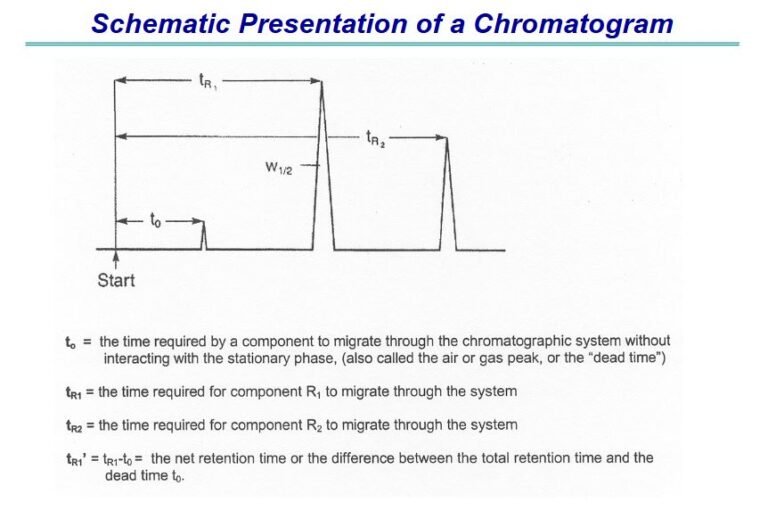
Specifically, section 506A of the FD&C Act and § 314.70 of FDA regulations provide for the following reporting categories of changes to an approved application:
-
Major Change: a change that has a substantial potential to have an adverse effect on the identity, strength, quality, purity, or potency of a drug product as these factors may relate to the safety or effectiveness of the drug product. A major change requires the submission of a PAS and approval by FDA before distribution of the drug product made using the change.
- Moderate Change: a change that has a moderate potential to have an adverse effect on the identity, strength, quality, purity, or potency of a drug product as these factors may relate to the safety or effectiveness of the drug product. Depending on the nature of the change, one of the following two types of supplements must be submitted to FDA for a moderate change:
- a.CBE-30 supplement – Changes Being Effected in 30 Days : A CBE-30 supplement involves certain moderate changes that require the submission of the supplement to FDA at least 30 days before the distribution of the drug product made using the change.
- b.CBE-0 supplement – Changes Being Effected : A CBE-0 supplement involves certain moderate changes that allow distribution to occur as soon as FDA receives the supplement.
- Minor Change: a change that has minimal potential to have an adverse effect on the identity, strength, quality, purity, or potency of a drug product as these factors may relate to the safety or effectiveness of the drug product. The applicant must describe minor changes in its next annual report
Performance Goals for PAS Submissions:
| Submission Type | Goal |
| Standard PASs |
|
| Priority PASs |
|
| Standard PAS Major Amendments |
|
| Priority PAS Major Amendments |
|
| Standard and Priority Minor PAS Amendments |
|
Is Inspections for PAS Submissions ?
As outlined above, the goal date for a PAS depends on whether the PAS requires an inspection. If a PAS does not require an inspection, the goal date is either 4 or 6 months from the date of submission; but if a PAS requires an inspection, the goal date is either 8 or 10 months from the date of submission.
For more details refer ANDA Submissions — Prior Approval Supplements Under GDUFA Guidance for Industry.
Specifically, this guidance describes how the GDUFA performance goals apply to:
- A PAS subject to the refuse-to-receive (RTR) standards
- A PAS that requires an inspection
- A PAS for which an inspection is not required
- An amendment to a PAS
- Other PAS-related matters