Purpose: To define the procedure for handling of market complaints.
- Scope: This guideline is applicable to the complaints of drug (formulation) products.
- Reference & attachments:
- References:
-
-
- in-house procedure.
- 21 CFR, Part 314.80
-
- Attachments:
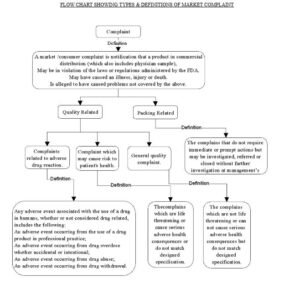
- Flow chart showing types and definition of market complaint. – Attachment I
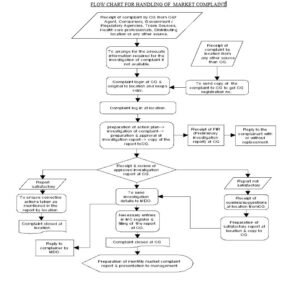
- Flow chart for handling of market complaint. – Attachment II
- Format of market complaint register. – Attachment III
- Format of Preliminary Investigation Report (PIR). – Attachment IV
- Format of action plan for investigation of market complaint. – Attachment V
- Examples of market complaint with guideline for investigation – Attachment VI
- Format for the investigation of market complaint. – Attachment VII
- Responsibility:
- Corporate Quality:
- To receive & log in the market complaint in market complaint register.
- To collect the adequate information required for complaint investigation if not available.
To send complaint to the concern location for the investigation.
- To receive market complaint investigation report or Preliminary Investigation Report
(PIR) from location.
- To review the investigation report.
- To send the investigation details to MDO/ Complainant / Distributing location.
- Quality Assurance:
4.2.1 To prepare or update market complaint SOP based on this guideline.
4.2.2 To log in the complaint in market complaint register.
4.2.3 To prepare PIR in case of quality related complaint where investigation may take time.
4.2.4 To prepare action plan for investigation of quality related complaint & to get it approved.
4.2.5 To conduct market complaint investigation as per action plan.
4.2.6 To prepare investigation report.
4.2.7 To get it reviewed and approved from concern persons.
- Quality Control:
- To monitor the analysis of complaint sample & / or control sample as per approved
action plan.
- To provide analytical results or any other data with conclusion.
- Production Head:
- To cooperate QA in the process of investigation as per the action plan.
- To review the investigation report.
- Head – Quality:
- To receive complaint from inhouse or any other source & to handover to QA for
investigation.
- To approve the action plan/to provide guidance for the investigation of market
complaint.
- To approve the investigation report.
- Head- Location:
- To approve the action plan/to provide guidance for the investigation of market
complaint.
- To approve the investigation report.
- Distributing location:
- To file complaints received of the products distributed by the distributing location and send it to the parent company for investigation.
- To receive investigation report from the parent company and take further action as per it’s location SOP.
- MD office:
- To arrange for replacement sample in case of genuine complaint as and when required with copy
to CQ.
- To prepare & send reply to customer/doctor as and when required with copy to CQ.
- Definition of terms
- Complaint:
A market /consumer complaint is notification that a product in commercial distribution (which also includes physician sample),
- May be in violation of the laws or regulations administered by the FDA.
- May have caused an illness, injury or death.
- Is alleged to have caused problems not covered by the above.
- Adverse drug experience:
Any adverse experience associated with the use of a drug in humans, whether or not considered drug related, includes the following:
- An adverse experience occurring from the use of a drug product in professional practice;
- An adverse experience occurring from drug overdose whether accidental or intentional;
- An adverse experience occurring from drug abuse;
- An adverse experience occurring from drug withdrawal and
- Any failure of expected pharmacological action.
- Life threatening adverse drug experience:
Any adverse drug experience that places the patient, in the view of the initial reporter, at immediate risk of death from the adverse drug experience as it occurred, i.e. it does not include an adverse drug experience that, had it occurred in a more severe form, might have caused death.
- Serious adverse drug experience:
Any adverse drug experience occurring at any dose that results in any of the following outcomes:
Death, a life threatening adverse drug experience, in patient hospitalization.
OR
Prolongation of existing hospitalization, a persistent or significant disability/incapacity, or a congenital anomaly/birth defect.
- Disability:
A substantial disruption of a person’s ability to conduct normal life functions.
- Unexpected adverse drug experience:
Any adverse drug experience that is not listed in the current labeling for the drug product. This includes experiences that may be symptomatically and path physiologically related to an event listed in the labeling, but differs from the event because of greater severity or specificity.
“Unexpected” as used in this definition, refers to an adverse drug experience that has not been previously observed (i.e. included in the labeling) rather than from the perspective of such experience not being anticipated from the pharmacological properties of the pharmaceutical product.
- Procedure:
- Source of complaint:
A complaint may be received from C & F agent, consumers, government/regulatory agencies, trade sources & health care professionals or any other source.
- Types of complaints: Refer attachment – I.
- Procedure for handling of complaints– Quality Related:
- Examples of quality complaints.
- Ineffectiveness,
- Melt back of lyophilized cake,
- Foreign matter,
- Color change,
- Change in physical form,
- Leakage,
- Adverse drug experience/reaction.
- Life threatening adverse drug experience.
- Serious adverse drug experience.
- Unexpected adverse drug experience.
- Receipt & registration:
- On the receipt of the complaint from any of the source mentioned under point 6.1, CQ shall log in the complaint, shall give CQ registration number & shall forward it to concern location with copy to MDO, wherever applicable.
- On the receipt of the complaint from CQ or any other source necessary entries shall be made at the location in the market complaint register (refer attachment- III).
- If location receives the complaint from any other source than CQ, copy of the complaint shall be sent to CQ immediately after receipt to get CQ registration number.
- Investigation of the complaint:
- Preliminary investigation:
- In case of quality complaint where there is a risk to patient health, preliminary investigation shall be over within 48 hours (Two working days) from the date of receipt of the complaint & preliminary investigation report (PIR) as per attachment IV shall be sent to CQ within two working days.
- If the complaint is confirmed, further actions shall be taken as per product recall procedure of the location.
- Location QA shall attach the PIR with the complaint, which shall be part of investigation report.
- In-case of complaint related adverse drug event, procedure described under CQ guideline no. 102/07 shall be followed.
- Detail investigation:
- If the complaint is confirmed, investigation shall be continued to investigate the complaint in detail & completed within 30 days.
- In case of general quality complaint investigation shall be over within 30 days from the date of receipt of complaint if does not demand for any additional work.
- QA shall conduct the investigation with the help of concern departments. QA shall prepare an action plan (as per attachment-V) based on guideline provided in attachment VI & shall get it approved from production, quality & location head before starting investigation.
- QA shall arrange for the physical inspection of control sample if required & shall prepare report of the same, which shall be part of the investigation report.
- QA shall provide required quantity of control sample to QC for analysis as mentioned in the action plan if required.
- QA shall provide required batch manufacturing documents to production or QC for review & data collection.
- Production & QC shall provide required information (as mentioned in the action plan) to QA for the preparation of investigation report.
- In-case of complaint related adverse drug event, procedure described under CQ guideline no. 102/07 shall be followed.
- QA shall prepare investigation report as per attachment VII & shall get it reviewed and approved by the concerns.
- QA shall send copy of the investigation report to CQ along with necessary attachments like control sample physical inspection report, trial batch report, quality control analytical data (in process trend, FP trend, stability data, complaint sample analytical results, control sample analytical results, trial batch results) or any other data suggested in the investigation action plan and the samples for replacement, where applicable.
- CQ shall review the investigation report & ask for further investigation details if required.
- CQ shall send investigation details to the MDO / Complainant / Distributing location.
- In case of quality complaint related to adverse drug reaction, following investigation procedure shall be followed.
- Complaint reported by one complainer:
- Location shall investigate the complaint as per investigation procedure explained under point no. 6.3.3.1 onwards & sent investigation report to CQ.
- The package insert &/or product information literature of the product for the information regarding contraindications, dosing, route of administration, warning, any other precaution etc.
- The information regarding use of suspected drug product in combination with other drug products.
- The patient history.
- CQ shall review Investigation report received from location & shall send the investigation details to MDO / Complainant / Distributing location.
Reported by more than one complainer:
- If the adverse experience reported is associated with the use of a drug product from more than one patient then, CQ intimates distribution department to hold the batch (es).
- Location shall investigate the complaint as per investigation procedure explained under point no. 6.3.3.1 onwards.
- Location shall review following,
- The package insert &/or product information literature for the information regarding contraindications, dosing, route of administration, warning, any other precaution etc.
- The information regarding use of suspected drug product in combination with other drug products.
- The patient history.
- Investigation report received from location along with complaint or control sample analytical results.
- Based on the review of the investigation report received from the location and after consultation with medical department CQ shall decide on the further action or on requirement of product recall.
- If required location & CQ shall proceed for the product recall as per location SOP.
- Preventive actions:
- After completion of the investigation,
- If required FDD shall be involved/informed for the preventive action. Refer CAR guideline No. 00054/03R1
- CAR (Corrective action recommendations) shall be issued by QA to concerned person to monitor the corrective actions.
6.3.5 Reply to complainer:
- If required, MDO shall send initial reply with/without replacement sample to the complainer & send a copy to CQ.
- Subsequently, CQ shall send copy of the initial reply to the concern location for the record.
- After receipt of the investigation report, CQ shall send either investigation details or reply for complaint to MDO with copy to concerned location for record purpose.
Procedure for handling of complaints– Related to short/excess receipt or improper packing:
- On the receipt of the complaint from any of the source mentioned under point 6.2, CQ shall forward it to concern location.
- If the complaint is received directly by location, a copy of the same shall be sent to CQ.
- Location QA shall forward the complaint to packing head.
- Subsequently packing head shall investigate & discuss the complaint with concern, impart the training & document the same.
- In-case of shortages related complaint investigation, discussion & training shall be over within 90 days from the date of receipt of complaint.
- Packing head shall send a copy of the training record to QA.
- QA shall do product wise trending of the complaints to take corrective actions (like change in pack style or any other) to prevent such complaints.
- If required QA shall send investigation report to CQ.
- CQ shall forward the same to MDO/ Complainant / Distributing location, if required.
Procedure for handling of complaints – Quality Related
(For the products manufactured on contract where
location is contract receiver)
- Receipt & registration:
- On the receipt of the complaint from any of the source mentioned under point 6.2, CQ shall log in the complaint, shall give CQ registration number & shall forward it to concern location.
- On the receipt of the complaint from CQ or any other source necessary entries shall be made at the location in the market complaint register (refer attachment- III).
- If location receives the complaint from any other source than CQ, copy of the complaint shall be sent to CQ immediately after receipt to get CQ registration number.
- Investigation of the complaint:
- Preliminary investigation:
- In case of quality complaint where there is a risk to patient health, preliminary investigation shall be over within 48 hours (Two working days) from the date of receipt of the complaint & preliminary investigation report (PIR) shall be sent to the coordinator of the concerned party within two working days.
- If the complaint is confirmed, further actions shall be taken as per product recall procedure of the contract giver.
- Location QA shall attach the PIR with the complaint, which shall be part of investigation report.
- Detail investigation:
- If the complaint is confirmed, investigation shall be continued to investigate the complaint in detail & completed within 30 days.
- In case of general quality complaint investigation shall be over within 30 days from the date of receipt of complaint if does not demand for any additional work.
- QA shall conduct the investigation with the help of concern departments. QA shall prepare an action plan (as per attachment-V) based on guideline provided in attachment VI & shall get it approved from production, quality & location head before starting investigation.
- QA shall arrange for the physical inspection of control sample if required & shall prepare report of the same, which shall be part of the investigation report.
- QA shall provide required quantity of control sample to QC for analysis as mentioned in the action plan if required.
- QA shall provide required batch manufacturing documents to production or QC for review & data collection.
- Production & QC shall provide required information (as mentioned in the action plan) to QA for the preparation of investigation report.
- QA shall prepare investigation report as per attachment VII & shall get it reviewed and approved by the concerns.
- Reply to complainer:
- Complaints received for the products, which are manufactured on contract for the other companies shall be handled by the respective manufacturing location in consultation with the contract giver.
- QA shall send copy of the investigation report to concerned party along with necessary attachments like control sample physical inspection report, trial batch report, quality control analytical data (in process trend, FP trend, stability data, complaint sample analytical results, control sample analytical results, trial batch results) or any other data suggested in the investigation action plan.
- QA shall send copy of the investigation report to CQ along with all attachments to close the complaint.
- CQ shall send investigation details to the MDO / Complainant / Distributing location.
- Procedure for handling of complaints – (Related to short receipt or improper packing)
(For the products manufactured on contract where
location is contract receiver)
- Detailed investigation shall be done if required in consultation with the contract giver/his representative in addition to the procedure described under
- In case of quality complaint product recall procedure shall be followed as per SOP of the contract giver.
- Procedure for handling of complaints – (Related to quality and packing).
(For the products manufactured on contract, where
location is contract giver)
- These complaints shall be handled through third party coordinator.
- Procedure for handling of complaints shall be same as motioned.