Author
"A seasoned professional with a Master's in Chemistry, the author brings more than two decades of expertise in Quality Control, Analytical Method Validation, and advanced QMS investigation. Their experience is globally validated by successful management of major regulatory audits, including those conducted by the US, EU, Brazil, and Russian authorities."
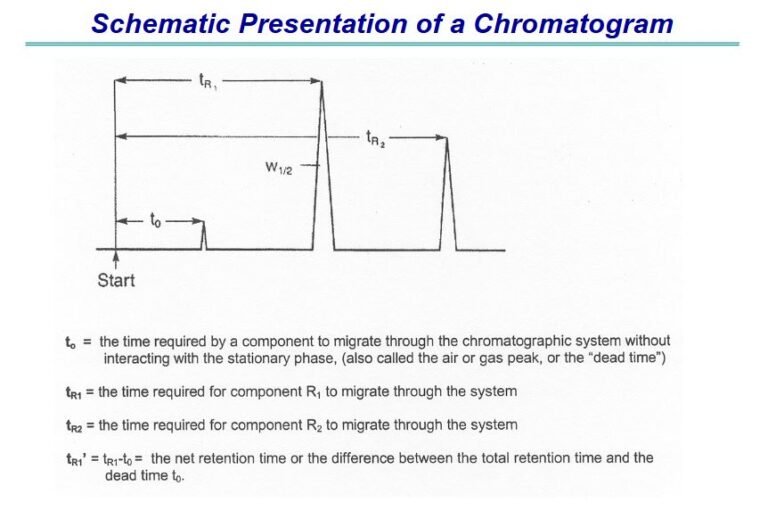
INTRODUCTION Chromatographic separation techniques are multistage separation procedures in which the components of a sample are distributed...
Column Life-Time : Q.: My column lasted only for about 100 injections. After that time, the peaks...
Cleaning Validation Manufacturing Facility & equipment – Manufacturing facility is nothing different, the same principles apply....
In Pharmaceutical Industries human error and its reduction is very bib challenge. During regulatory audit most of...
Transit (Transportation ) stability study is very important for Pharmaceutical Industries to export the medicines to other countries. After ...
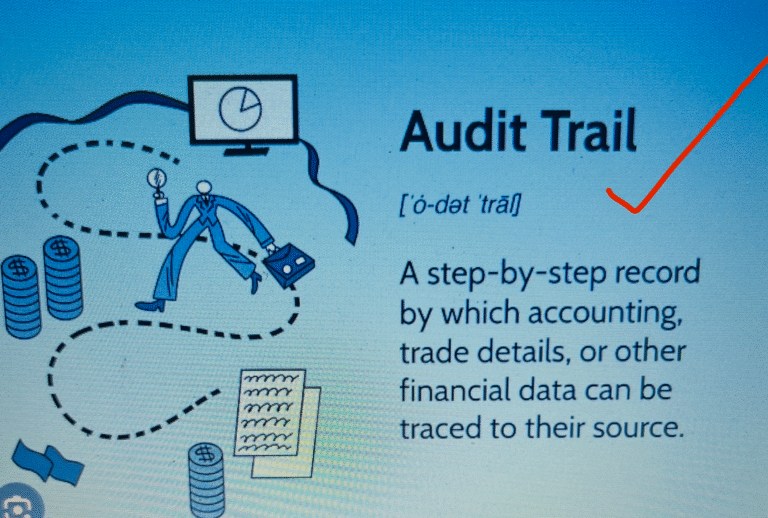
Audit Trail: “An audit trail (also called audit log) is a security-relevant chronological record, set of records, and/or destination and source...
Introduction: Cleaning Validation is very important topic in Pharmaceutical Industries. Our cleaning procedure and cleaning validation should...
Brief: In any pharmaceutical organization compliance of 21 CFR part 11 is very important and basic requirements...
As per ICH “significant change” the changes occur in the drug product during the stability study in Accelerated condition (ACC). In...