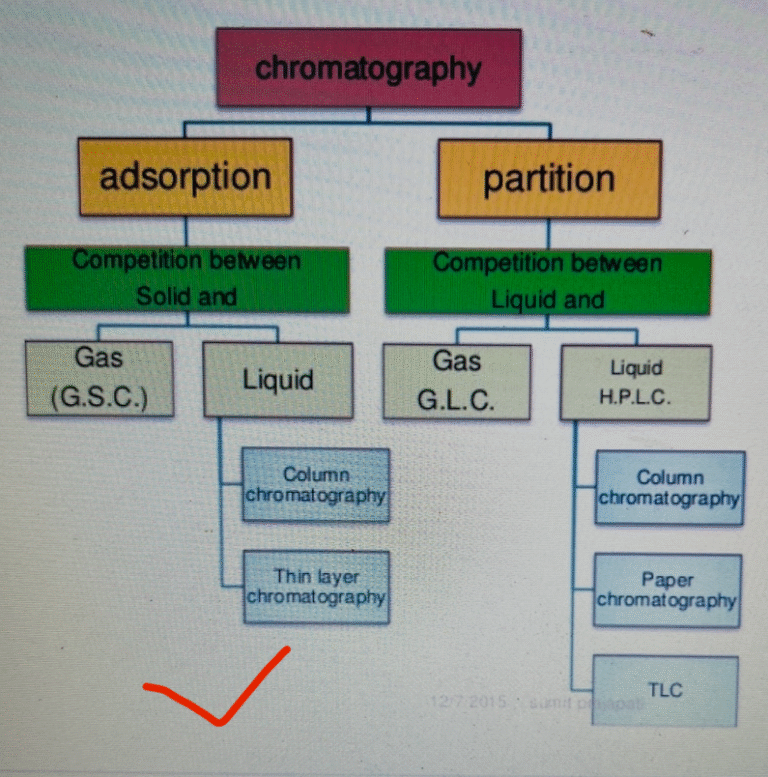
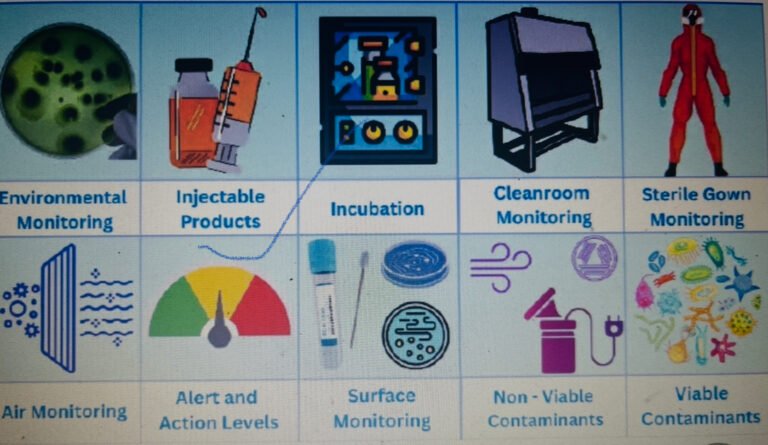
In pharmaceutical industries specially in Quality control laboratory, it is very difficult to decide that the event...
Author
"A seasoned professional with a Master's in Chemistry, the author brings more than two decades of expertise in Quality Control, Analytical Method Validation, and advanced QMS investigation. Their experience is globally validated by successful management of major regulatory audits, including those conducted by the US, EU, Brazil, and Russian authorities."
In Pharmaceutical Industries, in most of the cases investigator has fixed many CAPAs without identifying the actual...
In Pharmaceutical Industries, Lab Incident investigation and root cause analysis to identify the source of Extraneous peak...
For OOS, USFDA published a guidance in October-2006 and further updated in May-2022 & MHRA published OOS...
Few important Questions and Answer related to ANDA submission are as:– To which generic drug product submissions...
Specifically, section 506A of the FD&C Act and § 314.70 of FDA regulations provide for the following...
Area Brief of observation Observation Applicable to All Department Building access records showed an employee responsible...
S. No. Problem Probable Cause Recommended Action 1 Noise Injector contamination Clean the injector or replace linear...
1.0 OBJECTIVE To lay down procedure for operation and calibration of HPLC, Quaternary Gradient with...
During regulatory audit in Pharmaceutical Industry it is very important to know each and every person of...