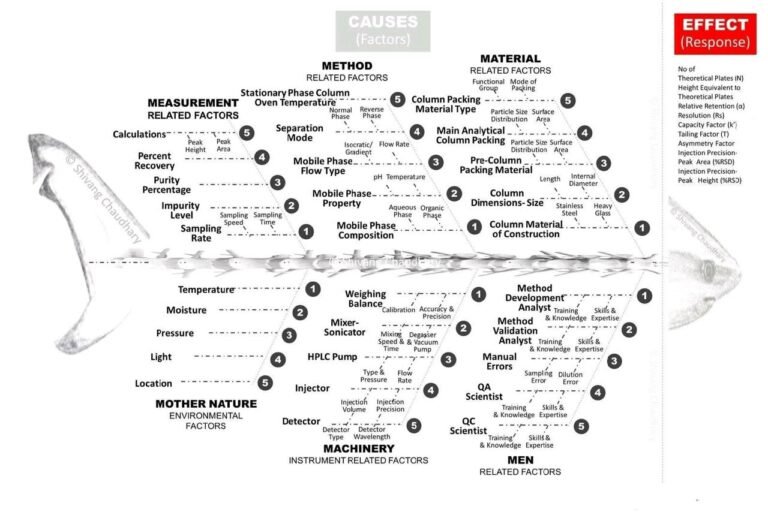
Chromatographic troubleshooting involves identifying and resolving problems that affect the quality, reliability and reproducibility of result....
Blog
1.0 OBJECTIVE: To lay down a procedure for investigation and evaluation of out-of-specification test results obtained...
1. Purpose: To define the procedure for stability studies. 2. Scope: This guideline is applicable to...
In Pharmaceutical Industries the Comparative Dissolution Profiles (CDP) is very important study. For CDP study enovator sample...
In Pharmaceutical Industries, it’s is very important to know the guidance for significant figure and rounding of...
Root cause analysis is a systematic process for identifying the true reason for a problem, not just...
In Pharmaceutical Industries generally following investigation tools are used by investigator- Different Investigation tools: A-5W and 1-How:...
Form FDA 483: USFDA declare an audit of any Pharmaceutical plant after ANDA filling or schedule GMP...
In Pharmaceutical Industries Related Substances test analysis is very critical, particularly when the HPLC method analysis is...
Many Pharmaceuticals organization received 483 due to selection of improper investigation tools. For example, system suitability is...