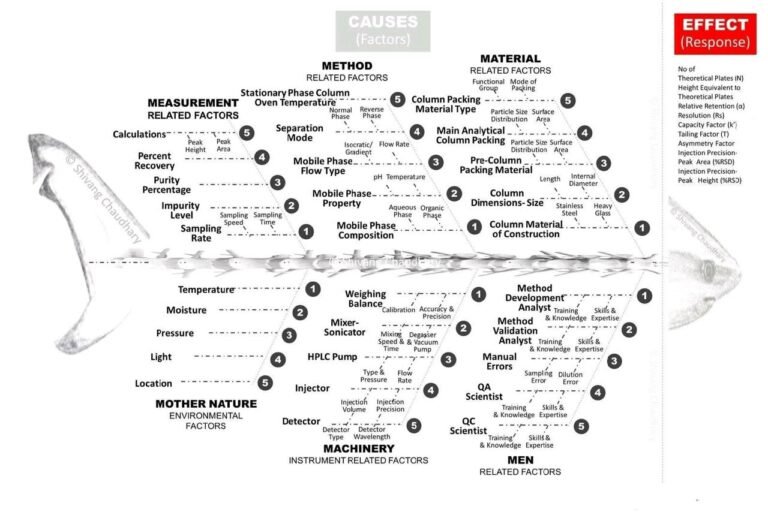
In Pharmaceutical Industries generally following investigation tools are used by investigator- Different Investigation tools: A-5W and 1-How:...
Mfg
Form FDA 483: USFDA declare an audit of any Pharmaceutical plant after ANDA filling or schedule GMP...
Many Pharmaceuticals organization received 483 due to selection of improper investigation tools. For example, system suitability is...
In Pharmaceutical Industries, Lab Incident investigation and root cause analysis to identify the source of Extraneous peak...
During regulatory audit in Pharmaceutical Industry it is very important to know each and every person of...
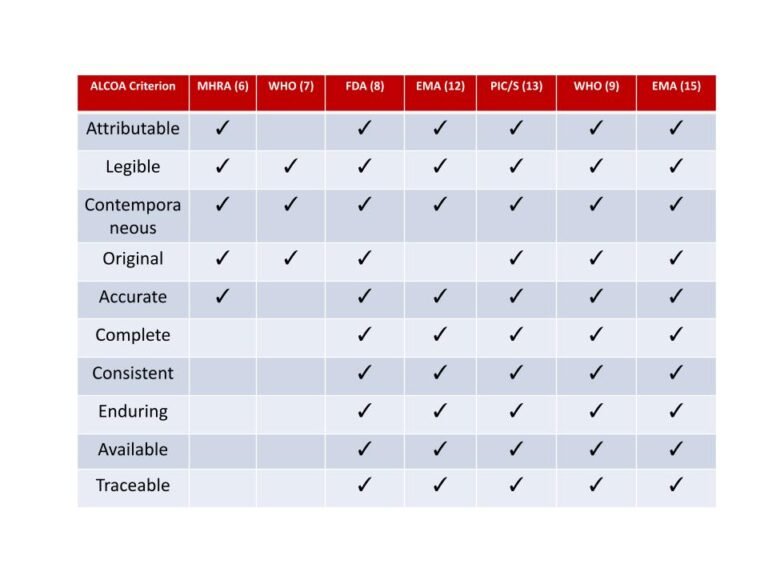
In Indian Pharmaceutical Industries, Data integrity is the favorite topic for regulatory auditors. Many organization had received...
In Pharmaceutical Industries OOS investigation and root cause identification is very important topic. If you are not...