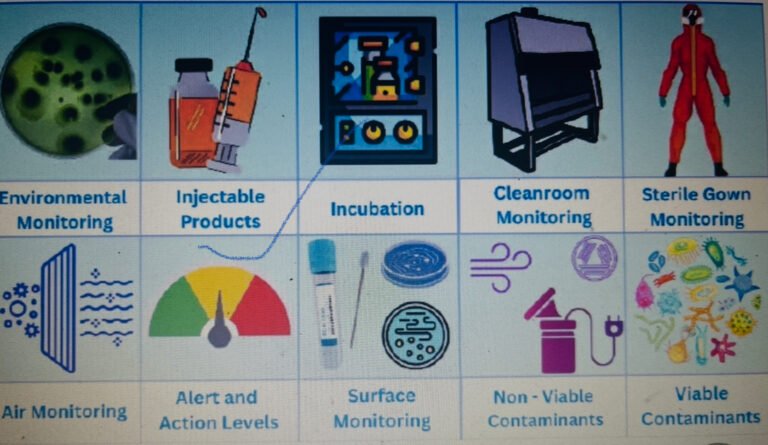
In Pharmaceutical Industries, Lab Incident investigation and root cause analysis to identify the source of Extraneous peak...
QA
For OOS, USFDA published a guidance in October-2006 and further updated in May-2022 & MHRA published OOS...
During regulatory audit in Pharmaceutical Industry it is very important to know each and every person of...
In pharmaceutical industries timely file a FAR for marketed batch is very important. Many companies got USFDA...
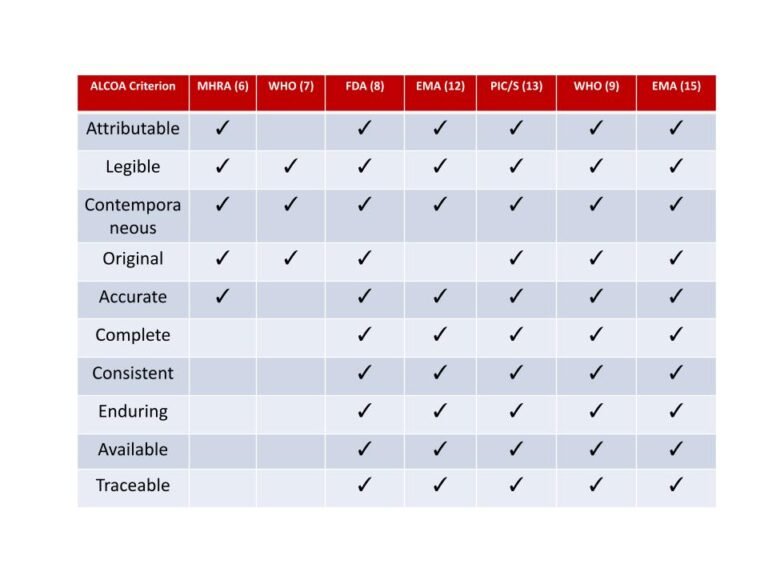
In Indian Pharmaceutical Industries, Data integrity is the favorite topic for regulatory auditors. Many organization had received...
In Pharmaceutical Industry, OOS investigation and root cause identification is very important topic. If you are not...
Case: OOS observed in Assay test. (Single preparation test and duplicate injections from same vial) During investigation...
In Pharmaceutical Industries OOS investigation and root cause identification is very important topic. If you are not...
In Pharmaceutical Industries OOS investigation and root cause identification is very important topic. If you are not...
Case: OOS in Water Content Test Here we will discuss about an OOS of destructive test (Water...