Cleaning Validation
Manufacturing Facility & equipment –
Manufacturing facility is nothing different, the same principles apply. We need to clean the Facility & Equipment, before and after the use.
Cleaning of the Facility and Equipment isrequired to avoid Contamination. This is the
very first requirement of GMP.
Why and When to validate?
- Cleaning Process Validation is required to establish documented evidence which provides a high degree of assurance that a selected cleaning procedure(s) will consistently clean the articles (equipment, facility, utensils, pipelines, components, heat exchangers, mills, filters, pumps, valves, etc.) meeting predetermined specifications of low level of acceptable contaminant(s).
- First we select appropriate cleaning procedure.
- Once the cleaning procedure is selected and finalized, one needs to validate it according to the “ICH Guidelines”.
THE QUESTION
- Which Step’s cleaning should be Validated?
- Which Equipment should be selected?
- Which Product(s) should be selected?
- What is the “Worst Case”?
- What should the Validation Protocol include?
- How to Develop & Select cleaning procedure?
- How to select Cleaning Material & Tools?
- How to arrive at “Acceptable Cleaning Levels”?
- What is the “Safety Factor”?
- What Sampling methods should be used?
- How to select Analytical Method(s)?
- How to Continuously Monitor &
- Conclusion
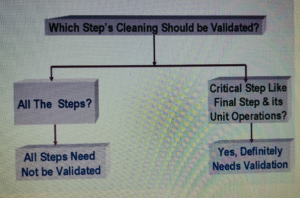
Which Step should be Validated?
- Cleaning procedures should normally be validated.
- Cleaning validation should be directed to situations or process steps where contamination or carryover of materials poses the greatest risk to Product quality.
- Identify the situations and critical process steps, where there is a risk of contamination or carryover of materials to the final product.
( e.g., Dryer, Blender, Filling Machine cleaning is most critical)
- Selection of Step for Cleaning Validation
Which Step’s Cleaning Should be Validated?
The steps of unit operations like sifting, , filtration, drying, milling, transferring, blending, weighing,Filling, packaging and labeling etc., of the Process are the most critical, so the cleaning procedure used for equipment for these steps should be validated.
Type of Cleaning
- Type-1: Batch to Batch of same product from lower potency to higher potency
- Type-2: Batch to Batch of same product from higher potency to lower potency.
- Type-3: Product to Product.
Miscellaneous type:
- Cleaning after Maintenance
- Cleaning after accidental contamination
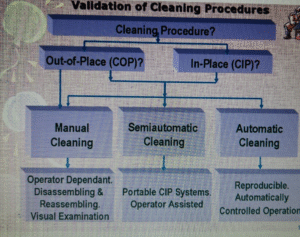
VALIDATION PROTOCOL
Cleaning Procedures
How to Select or Develop Cleaning Procedure?
- It should be as simple as possible and should be reproducible (Validation required).
- It should be able to remove the residue to the level of acceptance limit.
- It should have Cleaning frequency.
The time gap from completion of manufacturing
process to the starting of cleaning process is
Important. Time lapse can make cleaning difficult.
Cleaning Materials
How to select Cleaning Agent(s)?
- Should be able to remove contaminant to the accepted level.
- Should not be retained as a contaminant.
- Should be available with specifications.
- Analytical method with low level of detection.
Examples of Cleaning Materials & Tools
- Water, Steam, Compressed air, Solvents,
Acids, Alkalis, Detergents, Scrubbing agents, etc.
- Brushes, Dusters, Sponges, Mops, Mirrors,
CIP systems or equipment.
- How to establish effectiveness of cleaning?
The success or the effectiveness of the cleaning procedure is determined by an estimation of residues left after cleaning and limiting them to acceptable levels.
Acceptable Cleaning Levels
How to arrive at acceptable levels?

Which Equipment Should be Selected?
If various Products are manufactured in the same equipment and the equipment is cleaned by the same process, a representative Product can be selected for the cleaning validation.
- How to Select Equipment, Product & Process for Cleaning Validation?
- As stated before, decide which step of the process needs to be validated and then select the equipment & product(s), process and analytical method(s) for equipment cleaning validation.
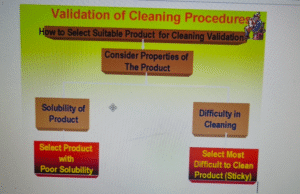
Which Products Should be Selected?
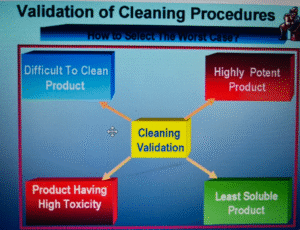
The selection (product or products) should be based on the solubility and difficulty of cleaning and the calculation of the residue limits based on potency, toxicity, and stability.
- Based on the list of the products manufactured, the “Product Matrix” should be prepared. The selection of the product(s) for cleaning validation should be based on certain principles like product’s properties and logic.
Establishment of Limits for Residue
The rational for determination of cleaning limits and acceptance criteria should be:
- Practical in the same sense that the limits chosen should be appropriate for the actual cleaning situation to be validated.
- Verifiable by some means of detection.
- Achievable by the analytical methodology available for the specific product.
- Acceptable to the regulatory authorities.
Sampling
Swab sampling may be impractical when product contact surfaces are not easily accessible due to equipment design, and/or process limitations (e.g., inner surfaces of hoses, transfer pipes, reactor tanks with small ports or handling toxic materials, and small equipment).
Analytical Methods
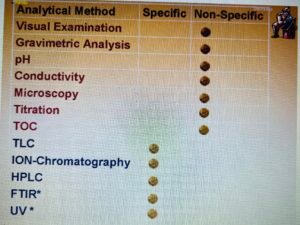
- Validated analytical methods having sensitivity to detect residues or contaminants should be used. The detection limit for each analytical method should be sufficiently sensitive to detect the established acceptable levels of the residue or contaminant. The method’s attainable recovery levels should be established.
- Specific and non-specific analytical methods are available.
Validation of Cleaning Procedures
Microbiological aspect
Equipment cleaning/sanitization studies should address microbiological and endotoxin contamination for those processes where there is a need to reduce total microbiological count or endotoxins in the product, or other processes where such contamination could be of concern

Cleaning Validation Approach
- At least three consecutive applications of the cleaning procedure for a single product on a single piece of equipment should be performed and shown to be successful.
- Challenging the cleaning procedure by deliberately contaminating the equipment in some way or by conducting the cleaning studies after normal use of the equipment may be carried out.
- Validation should be based on the worst-case scenario including challenges to the process.
- Standard operating procedures (SOPs) should be developed in sufficient details to ensure process consistencies.
- A cleaning validation protocol and final review report should be prepared.
Validation Protocol
The cleaning validation protocol should
describe:
- a) The equipment to be cleaned
- b) Cleaning procedures
- c) Cleaning materials (Agents & Tools)
- d) Acceptable cleaning levels (limits)
- e) Analytical methods (validated)
- f) The protocol should also indicate the type
of samples to be obtained and how they are collected and labeled.
Monitoring
Cleaning procedures should be monitored at appropriate intervals after validation to ensure that these procedures are effective when used during routine production. Equipment cleanliness can be monitored by analytical testing and visual examination, where feasible.
CONCLUSION
The cleaning validation program should be based on :
- Detailed cleaning procedure
- A good training program
- A validation protocol
- Validated chemical and microbiological methods
- A final report and auditing required to ensure compliance