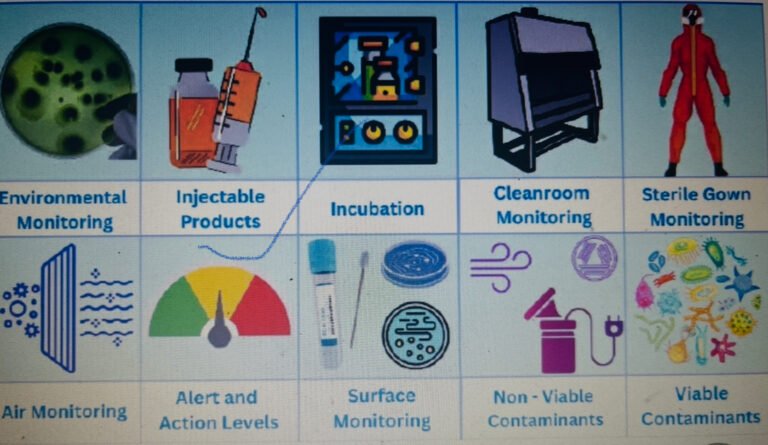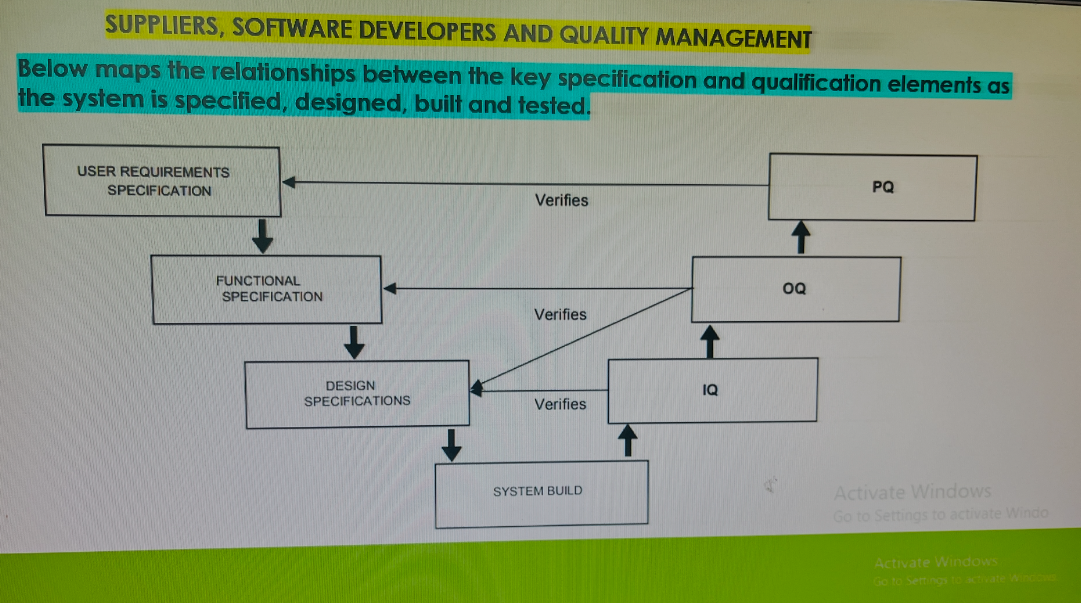
Brief:
The Basics of CSV Plan are as;
Scope:
This plan applies to all computerized systems implemented at Torrent Pharmaceuticals Limited and its subsidiaries. It covers both off-the-shelf systems and customized systems developed for Torrent-specific processes. The plan ensures compliance with current international regulations and guidelines. Any changes in such documents or interpretations will be reviewed periodically, and necessary revisions will be made to this plan.
Policy Guidelines and References:
This document is based on recognized international regulatory and guidance documents, including:
- US FDA 21 CFR Part 11 – Rules for Electronic Records and Signatures
- EudraLex Volume 4 GMP, Annex-11: Computerized Systems
- PIC/S Guide to GMP for Medicinal Products – Annexure 11 for Computerized Systems (Published by PIC/S)
- GAMP 5: A Risk-Based Approach to Compliant GxP Computerized Systems
- GAMP Good Practice Guides, including:
- – A Risk-Based Approach to Operation of GxP Computerized Systems
- – A Risk-Based Approach to GxP Process Control Systems
- – A Risk-Based Approach to GxP Compliant Laboratory Computerized Systems
- Global Information Systems and Control and Compliance – ISPE Publications
- A Risk-Based Approach to Testing of GxP Systems
- IT Infrastructure Control and Compliance
- MHRA GMP Data Integrity Definitions and Guidance for Industry
Validation Approach
The Validation Master Plan describes the detailed activities and approach for validating computerized systems. This structured methodology ensures that computerized systems are implemented in a manner that complies with GxP requirements. To minimize risks, the plan follows the GAMP 5 methodology, which emphasizes a life-cycle approach to system development, implementation, and maintenance.
System categorization will be based on GAMP 5 software categories, which help determine the appropriate validation strategy. Where suppliers provide system documentation, the adequacy and current applicability of such documentation will be reviewed against this plan. Additional documents will only be created if supplier documentation is found insufficient.
21 Code For Federal Regulations Part 11 is a law that ensures organization implement good practices. Part 11 allows a company to implement computer systems that will highly increase th e efficiency of individuals, minimize the errors by identifying risks, and increase overall productivity of the organization.
The Code of Federal Regulations (CFR) contains the laws for each of the government agencies. Each title of the CFR addresses a different regulated area. Laws typically refer to records and approval signatures, which originally referred to paper documents and handwritten signatures. Part 11 allows any paper record to be replaced by an electronic record, and allows any handwritten signature to be replaced with an electronic signature.
While Part 11 is an essential and very successful law, there has been much controversy and misunderstanding about it. The law is less than three pages long and doesn’t give much detail about electronic records and signatures. Don’t be mislead by the almost 30 pages of preamble material that is not the law. Just go to the end of the Part 11 document and flip back three pages to the beginning of the law. Adding to the confusion is the rapid evolution of computer technology that has made 21 CFR Part 11 compliance a moving target.
Challenge test:
All positive as well negative challenge test should be performed at the time of installation and this should comply to it’s Pre-approved acceptance criteria.