Understanding the FDA 483
A Form FDA 483, often called an “inspectional observation,” is a notice given by the U.S. Food and Drug Administration (FDA) to a company’s management at the conclusion of a facility inspection. The form lists the conditions or practices that may violate the Food, Drug, and Cosmetic Act and related acts. Receiving a 483 indicates that the FDA inspector found something wrong at the facility.
The Investigation Misstep:
The scenario we described is a example of this. Let’s understand:
- Initial Failure: A laboratory test is performed, the result is OOS (Assay: 94.8% against the limit of 95.0% to 105.0%) and a key system suitability parameter also fails in the Relative Standard Deviation (RSD) for bracketing standards is out of limit. This indicates that the analytical system itself (the instrument, column, etc.) isn’t performing reliably.
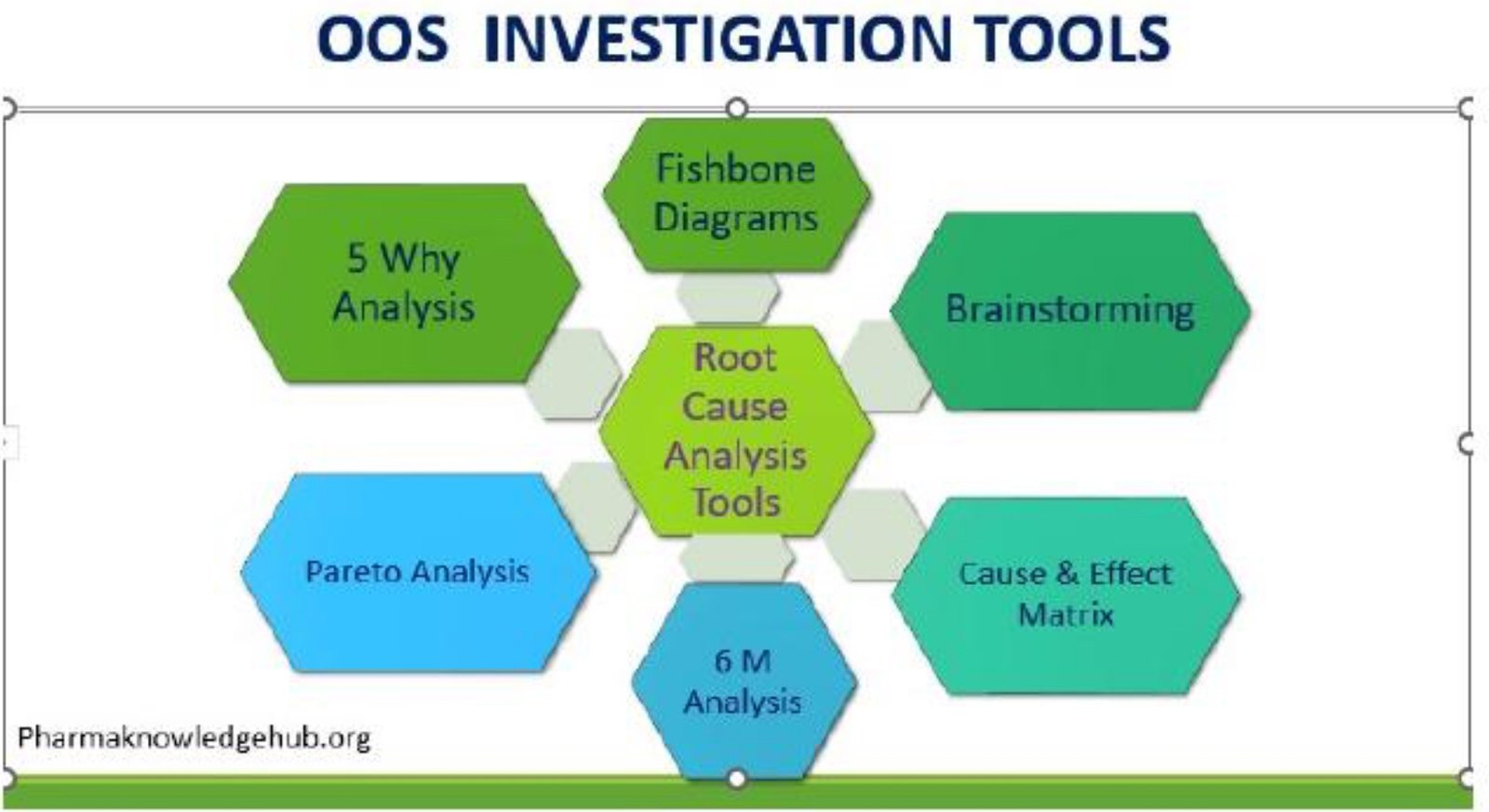
- The Wrong Tool: Instead of proceeding with an OOS investigation, the lab initiates a “lab incident” investigation. This is the critical mistake. An OOS investigation is a formal, rigorous process specifically designed to determine the root cause of a result that doesn’t meet its acceptance criteria.
- The FDA’s View: The FDA may considers this a serious issue. By avoiding the OOS procedure, the company is perceived as trying to hide or minimize a product quality issue. This may consider a violation of Current Good Manufacturing Practices (cGMP). It shows a lack of control and integrity in the quality system, which can lead to a 483, and potentially halt production and distribution.
Proper Procedure
The correct path is straight forward:
- OOS Investigation: If the system suitability is failed and run also produced an OOS result for the product, the OOS procedure must be initiated immediately. This is not optional. The investigation must determine if the OOS result is valid and, if so, what its root cause is.
- Lab Incident (When Appropriate): A lab incident is appropriate only when a non-conformance occurs that does not lead to an OOS or Out-of-Trend (OOT) result. For example, if a system suitability parameter fails, but there is no OOS result and the failure is determined to be an equipment malfunction, a lab incident investigation is the correct tool to address the equipment issue.
In essence, an OOS result always triggers an OOS investigation, regardless of other concurrent failures. We can’t use a less formal tool to address a serious product quality issue.