1-OBJECTIVE
To lay down a procedure for product recall.
2-SCOPE
This procedure is applicable for Product recall at xyz
3-RESPONSIBILITY
- QA Head or his/her authorized designee for notification and coordination of Product Recall procedure.
- Head Distribution/ Marketing or his/her authorized designee for issuance of recall notice to all stockiest, C&F Agents, Distributors, Wholesalers and Retailers to retrieve the stocks.
4.0 ACCOUNTABILITY
Plant Head.
5.0 PROCEDURE
5.1 Product Recall: Product Recall is removal of a specific batch or batches of the product from the market.
5.2 When any critical complaints or critical defects are observed during investigation of a major
complaint. Below of marketed batches of products,
Physician samples, Samples for Clinical trials shall be considered for product recall.
- Classification of Defects:
5.3.1 Class I defects:
Defects which shall be potentially life threatening or could cause a serious risk to the health of the user. For example-
- Products mix-up.
- Product not meeting regulatory specification.
- Chemical contamination with serious medical consequences.
- Mix up or use of wrong printed packaging materials.
- Adverse drug reaction or drug incompatibility.
- Class II defects:
Defects that could cause illness, but are not covered under Class I. For example
- Missing or incorrect information (leaflets or inserts).
- Microbial contamination of solids and other dosage forms.
- Physical changes in the product such as discoloration or precipitation.
2. Class III defects:
Defects that may not cause a significant hazard to health, but withdrawal shall be initiated for other reasons. For examples:
- Faulty over printing e.g. wrong or missing batch number or expiry date.
- Contamination e.g. microbial growth due to spillage/ damage/ breakage, dirt etc
- Advise of Drug Control Authority or Regulatory Authority.
- QA Head in consultation with Plant Head shall take the decision of Product Recall based on the critical complaints, critical defects observed during investigation in the major complaints or due to any reasons mentioned in point 5.3. Flow chart for Product Recall Procedure is mentioned in Annexure-V.
- QA Head shall classify the defects as per point 5.3 and action shall be taken accordingly.
- The procedure of Product Recall shall be completed within 10, 20 and 30 days for Class-I, Class-II and Class-III respectively (as described in point no. 5.3) from the date of complaint received in case of the product sale in India only.
- If the product to be recalled is exported, QA Head shall inform to the respective Regional Director/ Country Manager for giving the information to the Regulatory Agency and procedure of Product Recall shall be completed as per Regulatory rules of that country where it has been exported.
5.8 Logging and Notification of Product Recall:
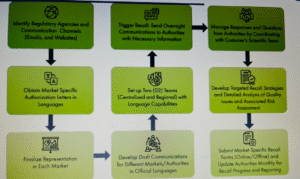
5.8.1 Once, recall of batch (es) of product has been decided, QA Head shall inform immediately to Marketing Head/ Distribution Head by using the fastest mode of communication (Phone, Fax, Email etc.) for stoppage of further sales and distribution of batch (es) of the product.
5.8.2 QA Dept. shall allocate the Product Recall Notification no. as described in point no. 5.8.3 and record the product recall details in Product Recall Log (Refer Annexure-I).
5.8.3 Product Recall Notification shall be numbered as PR/YY/NNN.
Where
PR stands for Product Recall.
YY stands for last two numbers of current calendar year.
NNN stands for serial number, starting from 001.
5.8.4 If the defect is Class-I or Class-II type, as described in point 5.3, QA Head shall inform immediately to the General public for alertness, stating that “not to consume the batch (es) of product” through media like newspapers, televisions and radio.
5.8.5 Marketing Head/Distribution Head shall immediately inform (by fastest mode of communication) to C&F agents, Stockiest, Distributors, Wholesalers and Retailers for stoppage of distribution and sale of the batch (es) of product in the market.
5.8.6 Marketing Head/Distribution Head shall have all the details (contact names/contact telephone numbers in working hours/out of working hours/) of all key personnel for immediate contact.
- List of Key personnel with contact numbers shall be prepared in Annexure – VI (List of Key personnel with their contact numbers) and updated as and when required.
- The batch (es) of product shall be kept under hold till the investigation is completed.
5.9 Investigation and Approval of Product Recall:
5.9.1 A detailed investigation regarding correctness of reported /identified defects shall be carried out by QA, FnD and Production, supported by Marketing and Distribution team. Opinion of Medical Advisor shall be sought to understand the health hazards of reported defects.
5.9.2 QA Head shall investigate to find out the cause of complaint and mention the findings and comments in Annexure-II.
5.9.3 The investigation shall be extended to others batches which might have been affected by the batch (es) under recall.
5.9.4 The investigation shall be completed within 7 days.
5.9.5 Plant Head shall recommend the action regarding Product Recall in Annexure-II.
5.9.6 Finally, QA Head shall summarize the comments from Plant Head, FnD, Production, Marketing/Distribution and Medical advisor in Annexure-II and recommend for further action.
5.9.7 Simultaneously QA Head shall collect all details of distribution of batch (es) of product and fill the data in Annexure-III.
5.9.8 QA Head and Plant Head shall write the comments and approve the product recall with sign and date in Annexure-III.
5.9.9 After approval of Product Recall, QA Head shall send the copy of Annexure-III to Marketing Head / Distribution Head for recall of the batch (es) of product and copy to the Regulatory authorities and Plant Head.
5.10 Product Recall:
5.10.1 After receipt of Annexure-III from QA Head, Marketing Head/ Distribution Head shall immediately inform to all C&F agents, Stockiest, Distributors, Wholesalers and Retailers etc. to retrieve the batch (es) of product.
- All retrieved and unsold stocks including physician’s samples of the batch (es), shall be quarantined in secure area with lock and key and marked as “RETURNED PRODUCTS” by the concerned In-charge.
- The Returned products shall be identified and stored separately in secured and segregated area with lock and key in warehouse as per the current version of SOP No. SQA- 025, till final decision on its fate.
- A copy of all public alerts published in newspapers and video film of television shall be sent to the QA Head for record.
- After retrieval of batch (es) of the product, QA Head shall decide about the fate of recalled batch (es) of the product. If the recalled batch (es) of the product are to be destroyed, destruction shall be carried out as per the current version of SOP No. SQA-051.
- If the recalled batches (es) of the product are not destroyed, same shall be handled as per the current version of SOP No. SQA-025.
- After completion of all activities of product recall, QA Head shall prepare a Product Recall Summary Report (Refer Annexure-IV) and same shall be circulated to all concerned departments.
5.11 Mock / Dummy recall shall be conducted for checking the effectiveness of Recall Procedure.